 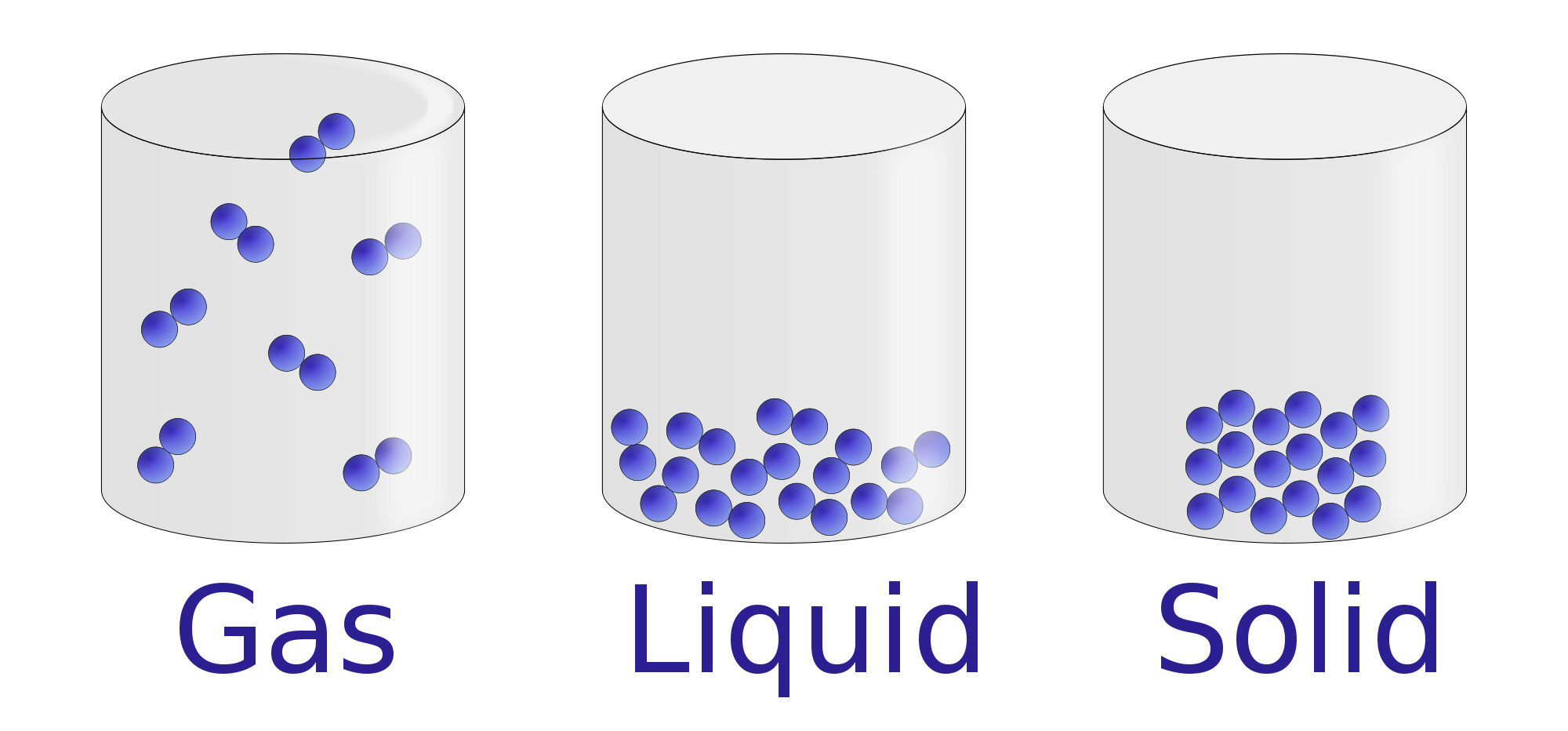
That is, in this chapter you will learn that water has stronger intermolecular forces than carbon dioxide, and that is why it is a liquid, while carbon dioxide is a gas. So why at ambient conditions is Carbon Dioxide (the heavier molecule at 44 amu) a gas, but water (the lighter molecule at 18 amu) is a liquid? Answer That is, shouldn't it be easier to vaporize the lighter molecule than the heavier one? The answer lies within the Intermolecular Forces, and if you review molecular shapes and polarity you will see that water is a polar molecule while carbon dioxide is non-polar, and this leads to different physical properties like the boiling and melting points. The problem is, there are often other factors, as indicated in the next question, on why is carbon dioxide a gas at ambient questions while water is a liquid? Note: This image is for the velocity distribution of a gas at a given temperature, and not a liquid, but the concept is valid, in that if all other things are equal, lighter molecules tend to move faster, and gas phase systems move faster than liquid. Look at the case of carbon dioxide and water (the next question deals with this). The problem is we need to identify the intermolecular forces that attract the molecules to each other, and if the lighter molecule had stronger intermolecular forces, this would be wrong. From our study of gases and the Kinetic Molecular Theory we learned that the average kinetic energy of a molecular system is proportional to the absolute temperature, and both molecules have the same average kinetic energy, and so at first thought, we would predict that the lighter molecule would tend to have the higher velocity (review Graham's Law of Effusion) and be easier to vaporize (see image below). The experiment also now holds the record for the coldest object we know of in space, though it isn't yet the coldest thing humanity has ever created.\)Īt a given temperature, would it be easier to vaporize a light molecule or a heavy molecule? Answer In July 2018, an experiment aboard the International Space Station cooled a cloud of rubidium atoms to ten-millionth of a degree above absolute zero, producing a Bose-Einstein condensate in space. Wieman, both of the Joint Institute for Lab Astrophysics (JILA) in Boulder, Colorado, and Wolfgang Ketterle, of the Massachusetts Institute of Technology, managed to make one, for which they got the 2001 Nobel Prize in Physics. Though the theory said such states of matter should exist, it wasn't until 1995 that Eric A. Bose-Einstein condensates break that rule. That in turn is one reason why chemistry works the way it does and one reason atoms can't occupy the same space at the same time. This is why when two electrons are in the same orbital, their spins have to be opposite so they add up to zero. Pauli (1900-1958) was an Austrian-born Swiss and American theoretical physicist and one of the pioneers of quantum physics.It says that fermions - the kinds of particles that make up matter - can't be in identical quantum states. 
Other states of matter all follow the Pauli Exclusion Principle, named for physicist Wolfgang Pauli. That's why the atoms in a Bose-Einstein condensate behave like "super atoms." When one tries to measure where they are, instead of seeing discrete atoms one sees more of a fuzzy ball. But cool the atoms to within billionths of a degree of absolute zero and some atoms begin to fall into the same energy level, becoming indistinguishable. This is why electrons, for example, have discrete "orbitals" that they have to occupy, and why they give off photons of specific wavelengths when they drop from one orbital, or energy level, to another. What the two found was that ordinarily, atoms have to have certain energies - in fact one of the fundamentals of quantum mechanics is that the energy of an atom or other subatomic particle can't be arbitrary. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
